Hospital oncology programs
Cancer and oncology programs within hospital systems that have not yet launched a CAR T-cell therapy program and need a clear path from readiness assessment through implementation.

acCELLapy is a specialized healthcare services company that partners with oncology providers, health systems, and biopharma to design, launch, and scale cell therapy programs. We bring together clinical, operational, financial, and quality expertise to help clients build programs that are safe, compliant, financially grounded, and ready to grow.
Cell therapy, and CAR T-cell specifically, has moved from a niche capability available at a limited number of centers since 2017 to a strategic requirement for oncology programs as indications expand and more patients become eligible for treatment. The question is no longer whether cell therapy will be part of your cancer service line. The question is how quickly and safely you can build a program that delivers these therapies to serve more patients.
Today, patient access remains constrained by limited program capacity, fragmented referral pathways, payor contracting complexity, and the difficulty of aligning clinical, financial, and quality requirements into one executable model. acCELLapy was created to help organizations close that gap and prepare for the next wave of advanced therapies.
We support organizations at different stages of cell therapy readiness, from first-time launch to multi-site expansion and post-launch optimization.
Cancer and oncology programs within hospital systems that have not yet launched a CAR T-cell therapy program and need a clear path from readiness assessment through implementation.
Independent medical oncology practices evaluating whether and how to offer advanced cell therapy safely, compliantly, and with a sustainable financial model.
Organizations that use shared SOPs and internal project management as a starting point, and partner with acCELLapy to add the specialized clinical, operational, and payor expertise needed to fully stand up and scale a safe, sustainable cell therapy program given their specific operating capabilities and market conditions.
Authorized treatment centers seeking to scale capacity, improve program profitability, optimize throughput, and extend capabilities to affiliated hospital networks so components of care can safely move closer to the community setting.
Biopharma organizations looking to expand market access, support site enablement, and strengthen provider readiness through education, training, and practical implementation support.
Every engagement is designed to answer a small set of critical questions for executives, clinicians, and program leaders such as:
We organize our work into connected pillars, so strategy, operations, quality, and economics are built together, not bolted on later or treated as things SOPs alone cannot solve.
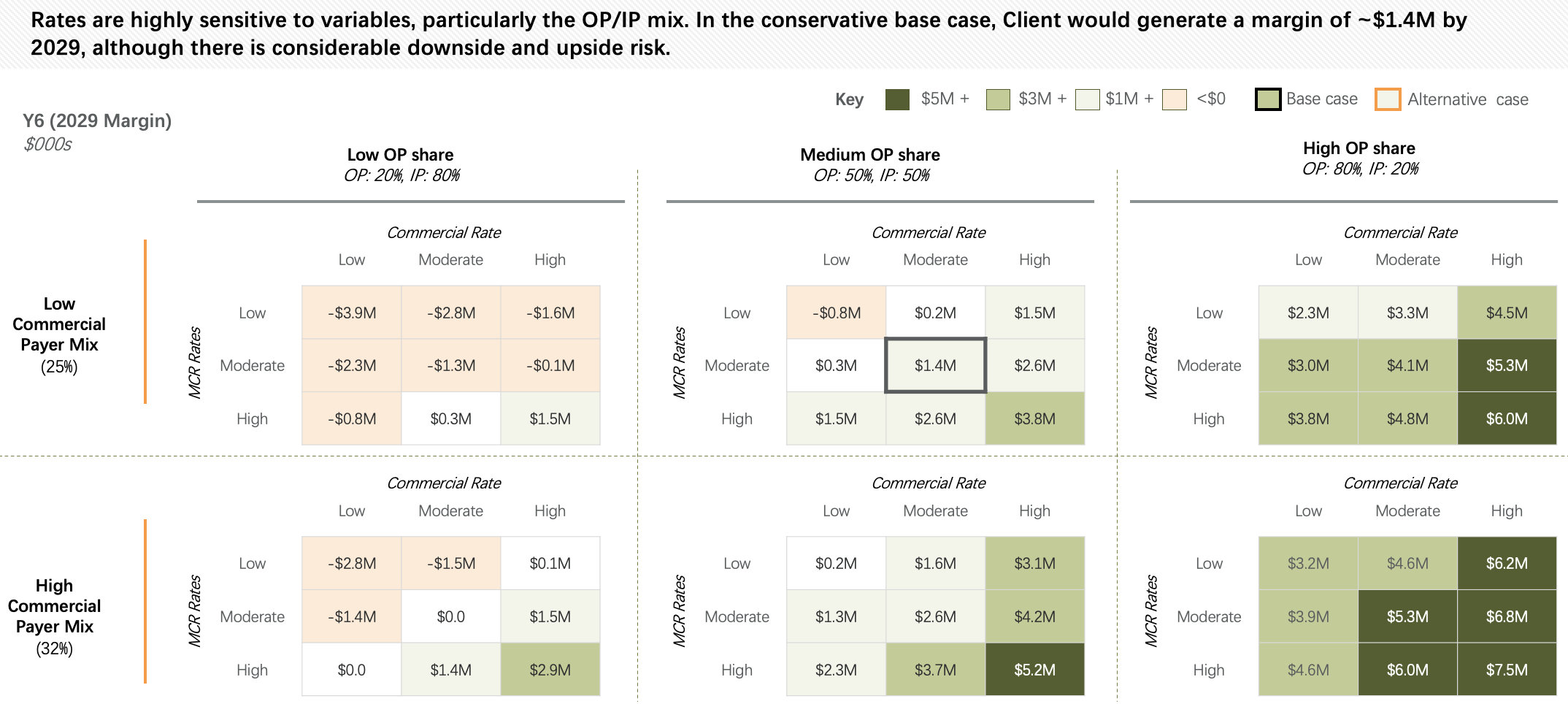
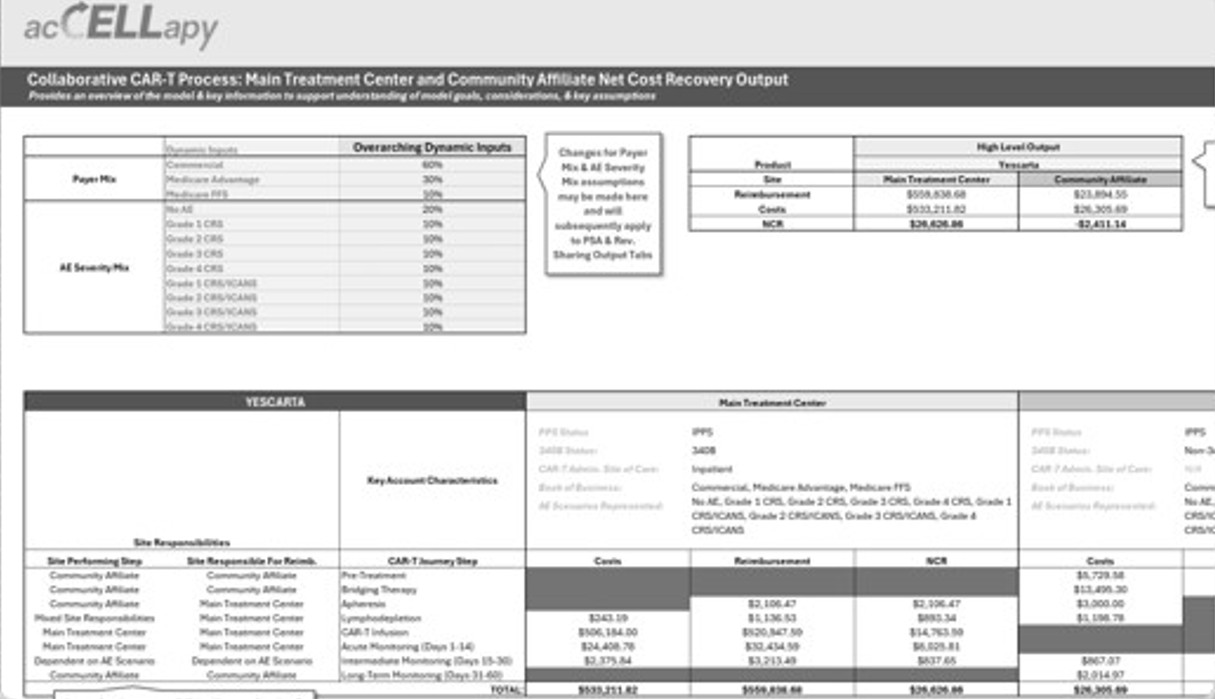
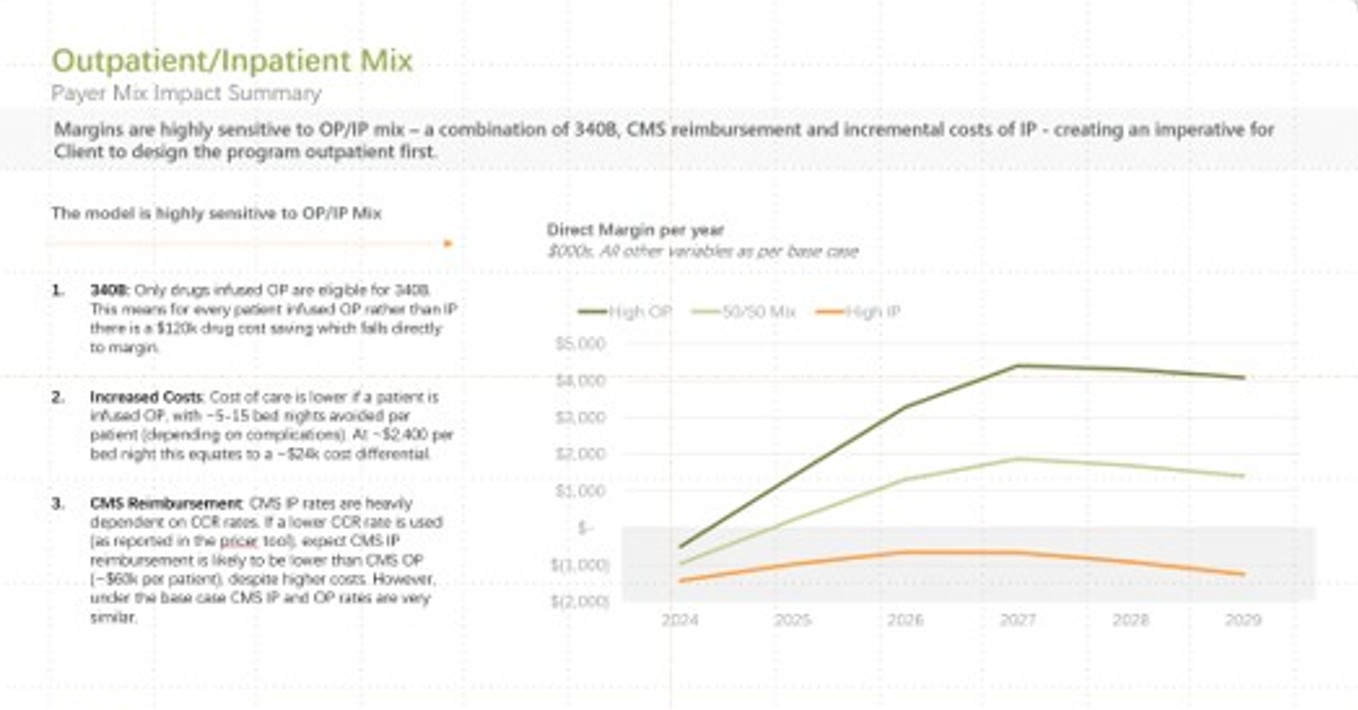
We assess market opportunity, referral dynamics, competitor activity, expected patient capture, and minimum viable program volume. We model bespoke cost per patient, forecast program economics, and help define the reimbursement and payer strategy, including likely case rates, to support a sustainable launch.


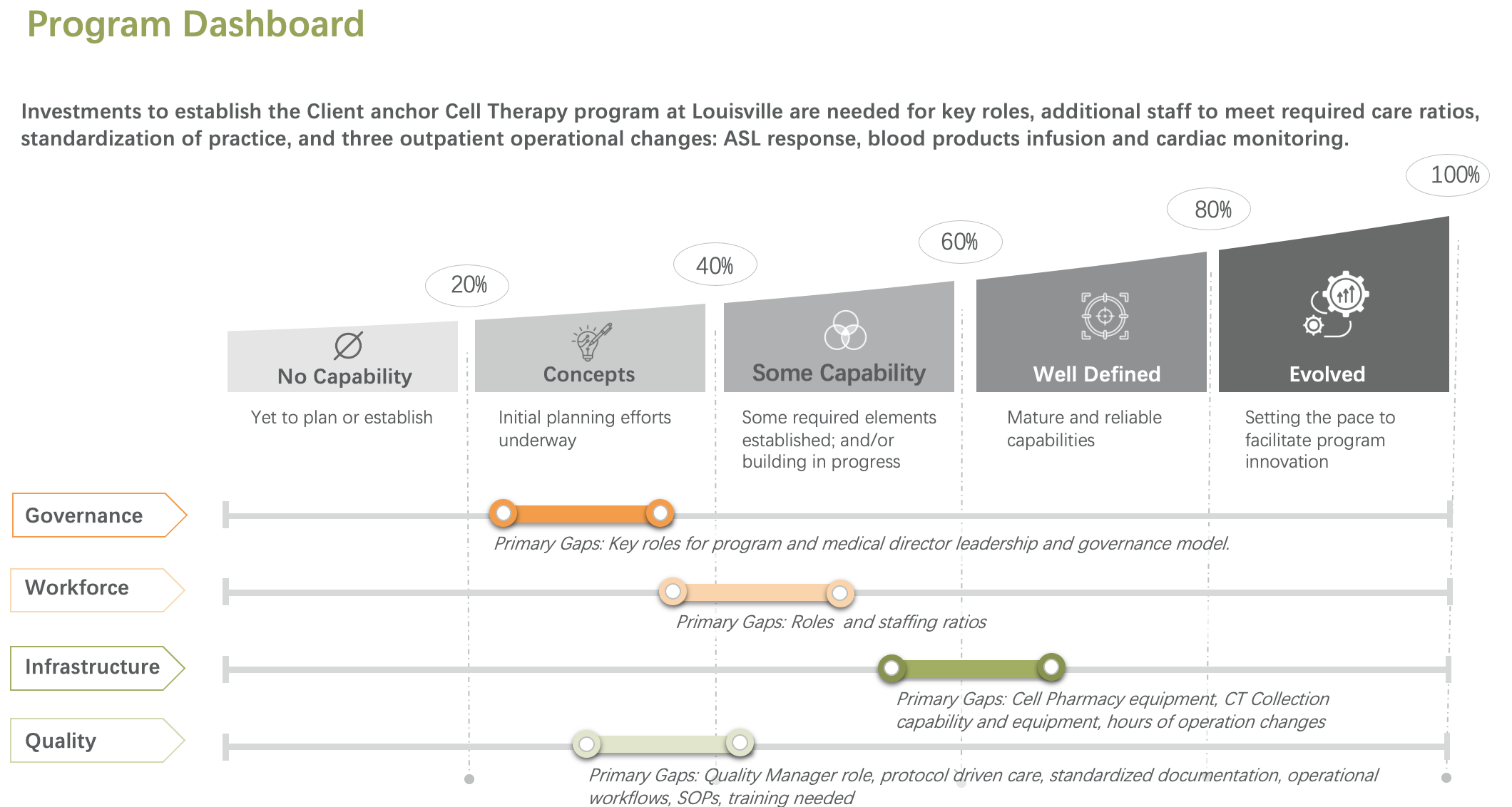
We evaluate your current-state capabilities across staffing, equipment, infrastructure, workflows, and patient management. This readiness and gap assessment clarifies whether your organization is prepared to launch, which gaps must be closed, and what sequencing is required to reduce execution risk.
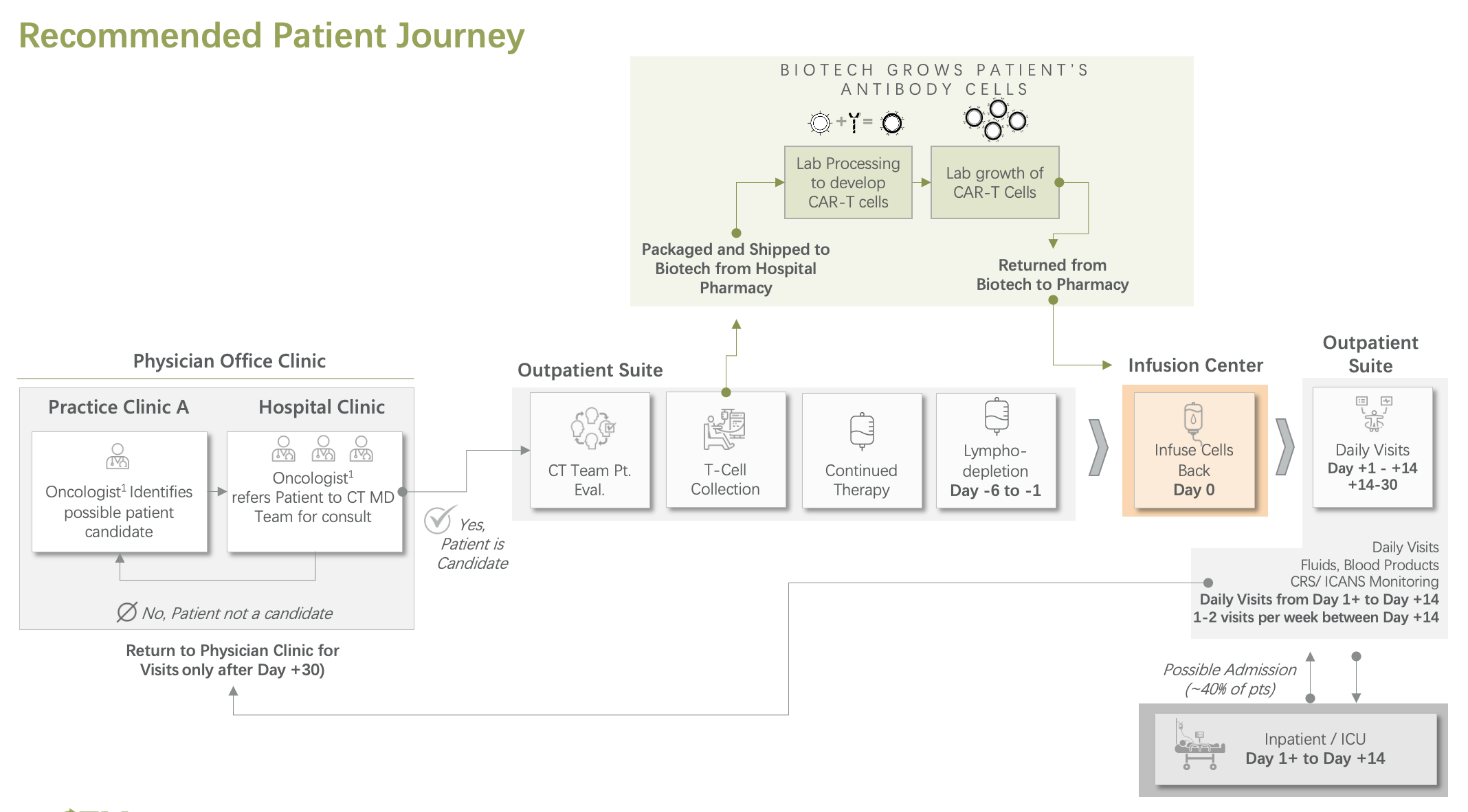
We translate strategy into a working program through a practical implementation roadmap and structured workstreams across the full patient journey, from patient identification and verification through apheresis, bridge therapy, lymphodepletion, Day 0 infusion, and post-infusion monitoring.
Many organizations begin with shared SOPs and internal project management, which can be a useful foundation. We build on that foundation by bringing hands-on clinical and operational expertise to design how the program actually runs in your environment, across teams, sites, and partners.
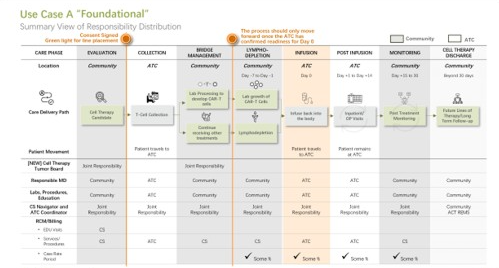

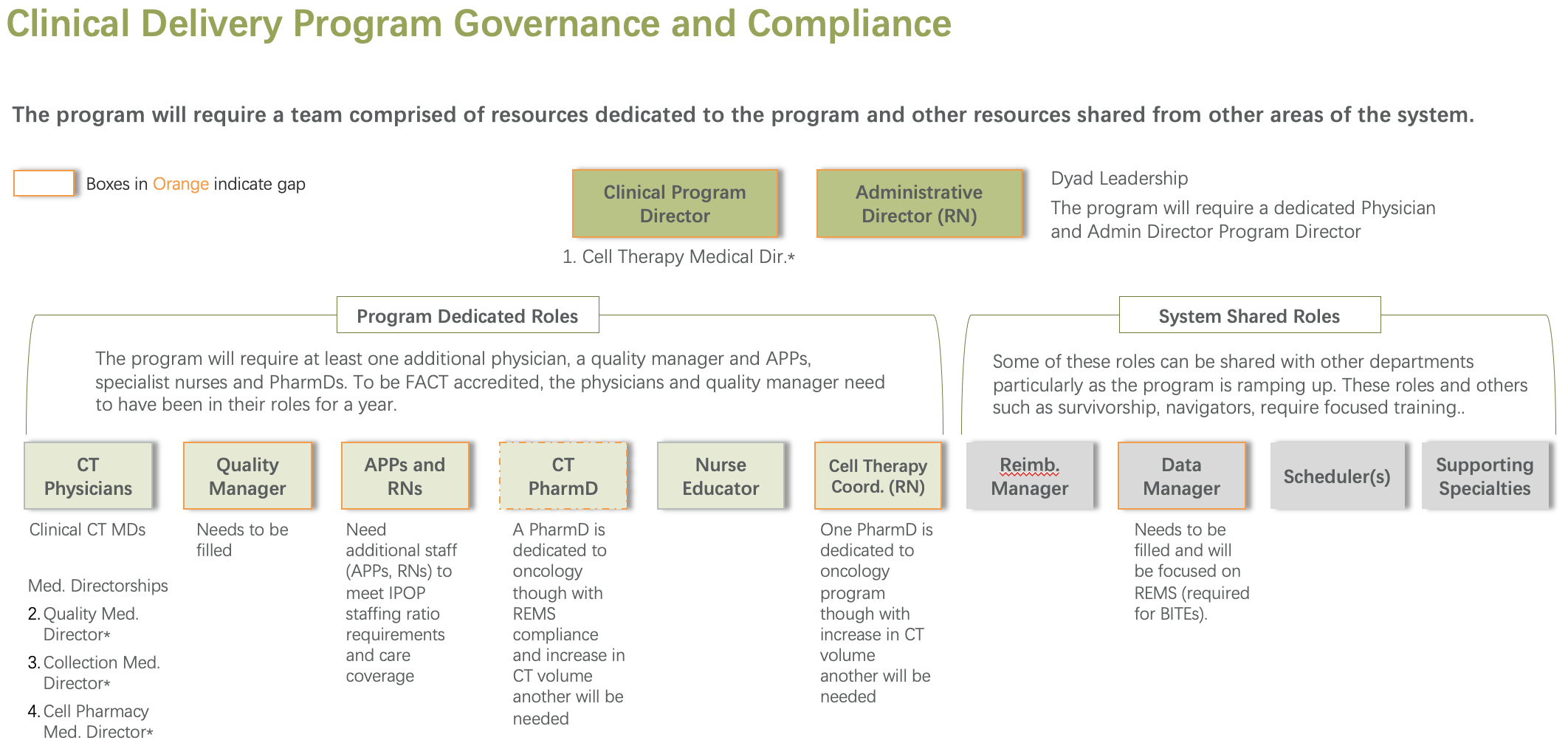
We support implementation with pre-developed program assets including draft SOPs aligned to FACT-related expectations, order sets, supply inventories, quality management plans, workflows, role clarity, and training for clinicians, nursing, and operational staff. These tools serve as implementation-ready building blocks that we refine and adapt together with your teams, not as plug and play documents.
We help you understand your likely cost per patient, design reimbursement strategies, and support payor contracting and case-rate negotiations. Our goal is to ensure that the program you build is both clinically sound and economically viable from day one.

After go-live, we remain engaged as a partner. We monitor program performance, review operational and financial results, track clinical outcomes, and support onboarding of new products as they enter the market. We also help you prepare for and maintain readiness against evolving expectations, including FACT-related requirements.
Most firms deliver recommendations, templates, and a workplan. Many organizations also try to move forward by sharing SOPs across sites and assigning internal project teams. acCELLapy complements those efforts by working elbow to elbow with your staff and bringing CAR T-cell physicians, nurses, and operational experts directly into the implementation effort.
Our model is built around pre-developed tools, job aids, SOPs, workflows, and implementation templates that have been refined through real-world experience. This allows clients to begin from a point of roughly 80 percent completeness and spend their time tailoring and validating, rather than reinventing the basics, so they can mobilize a program in months instead of years.
We integrate financial planning, program design, quality, payor strategy, and patient safety into one coherent operating model. Your program is grounded in real-world clinical and operational constraints, not just spreadsheets and slide decks, and shared documents are translated into daily practice.
Through established relationships across the cell therapy ecosystem, we connect clients to pre-vetted partners for services such as apheresis and patient monitoring. This gives organizations flexibility in how they build their model and can reduce the need to construct every component alone.
All eligible cancer patients should be able to access advanced cell therapy through high-quality programs closer to home.
Our mission is to prepare oncology organizations for the coming wave of cell therapy by building the capabilities, workflows, and operating models required to expand access safely, improve referral conversion, and fundamentally change the current treatment paradigm in which too few eligible patients receive these therapies.

Co-founder, acCELLapy
Beth A. Price, MBA is a co-founder of acCELLapy, LLC, where she leads strategy and services development to help U.S. health systems and community oncology programs design and launch cell therapy and CAR T treatment capabilities. A healthcare executive and strategist with deep experience across providers and life sciences, she specializes in therapy adoption, clinical research platforms, and oncology service line growth.
At acCELLapy, LLC, Beth partners with oncologists, pharmacy, finance, and operations leaders to translate complex cell therapy and CAR T requirements into practical business plans, care models, and workflows that expand patient access while supporting sustainable economics. She created acCELLapy as an advisory platform to address Cell Therapy Program readiness or barriers, and to extend novel therapies into community programs, where the majority of cancer patients receive care.
Earlier in her career, Beth built and led nationally recognized oncology research and care delivery platforms, including serving as CEO and Executive Director of the Vanderbilt-Ingram Cancer Center and cofounding the Sarah Cannon Research Institute. Beth led the Oncology Solutions consulting practice at The Chartis Group, advising cancer programs and practices nationwide on strategy, transactions, performance improvement, and research-enabled growth. These roles gave her a dual lens as a health system C-suite leader and consulting practice leader, informing her work at the intersection of cancer centers, community oncology networks, and life sciences sponsors.
Whether you are evaluating a first launch, scaling an existing center, or extending access across a broader network, acCELLapy helps you move from strategy to implementation with confidence.